Bayer HealthCare LLC, of Whippany, New Jersey Bayer Recalls 6 mL Size Afrin Original Nasal Spray Bottles Due to Risk of Seriou Recall 2026
Bayer HealthCare LLC, of Whippany, New JerseyIssued April 30, 2026
Bayer HealthCare LLC, of Whippany, New Jersey Bayer Recalls 6 mL Size Afrin Original Nasal Spray Bottles Due to Risk of Serious Injury or Illness from Child Poisoning; Violates Mandatory Standard for Child-Resistant Packaging — recalled per CPSC on 4/30/2026. Reason: The 6 mL nasal sprays contain an imidazoline, which must be in child-resistant packaging or meet the labeling requirements for non-complying packaging, as required by the Poison Prevention Packaging Act. The 6 mL nasal spray's packaging is not child-resistant nor bears the required labeling statement, posing a risk of serious injury or illness from poisoning, if the contents are swallowed by young children..
What to do right now
- 1
Stop using it
Take it out of rotation. Don't consume, wear, or operate.
- 2
Verify the match
Compare lot numbers, UPCs, and dates against your product.
- 3
Return or dispose
Follow the manufacturer's instructions to refund or safely dispose.
Verified source
U.S. Consumer Product Safety Commission
Summary of the official CPSC notice issued April 30, 2026. Last synced May 1, 2026. View on cpsc.gov
What's recalled
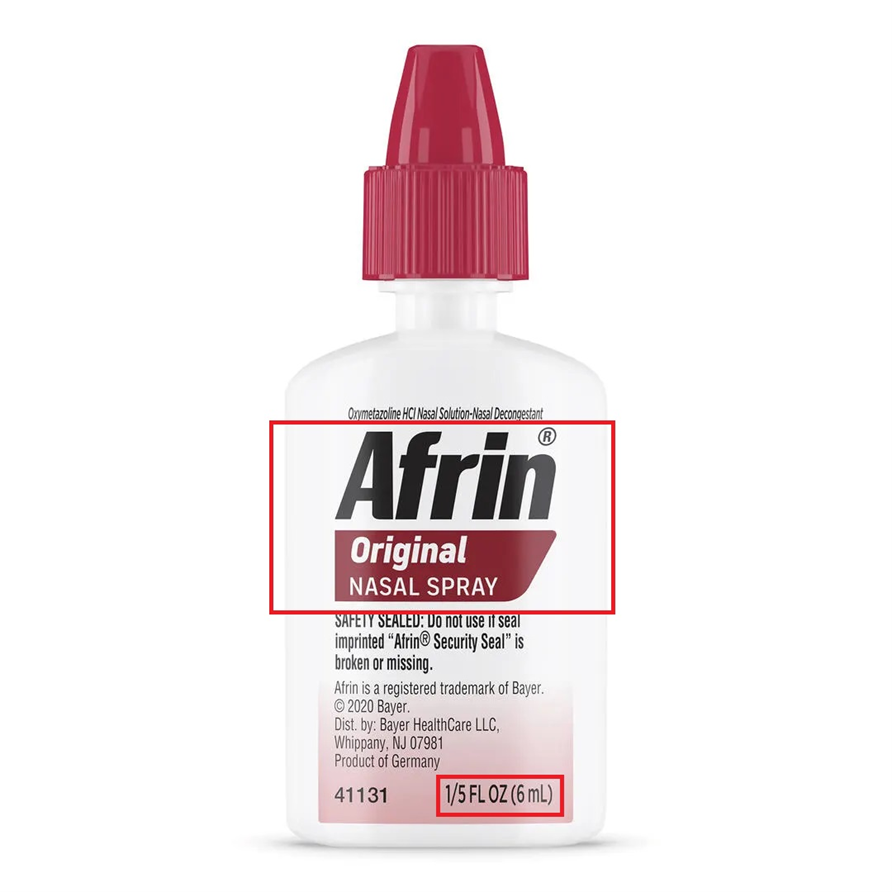
This recall involves unexpired Travel Size Afrin® Original Nasal Spray 6 mL bottles, with Lot numbers 230361, 240822, 241198, 250066, 250152, 250646, and 250831. These travel size bottles have "Afrin® Original Nasal Spray" and "1/5 FL OZ (6 mL)" printed on a label located on the front of the bottle. It is a six-digit number, followed by the expiration date in following format: "YYYYMMM". No other sizes of Afrin are included in this recall.
Why it was recalled
The 6 mL nasal sprays contain an imidazoline, which must be in child-resistant packaging or meet the labeling requirements for non-complying packaging, as required by the Poison Prevention Packaging Act. The 6 mL nasal spray's packaging is not child-resistant nor bears the required labeling statement, posing a risk of serious injury or illness from poisoning, if the contents are swallowed by young children.
Where was it sold1 state
View all questions for this recall →
More recalls like this4 related — by brand, contaminant, and category
Recent Baby & Kids recalls
- Alliance Chemical, of Taylor, Texas Alliance Chemical Recalls 1-K Kerosene Heater Fluid Containers Due to Risk of Se Recall 20262026
- CBC Group Recalls Stephan Baby Boo Bunnie and Friends Children's Toys Due to Ris Recall 20262026
- Build-A-Bear Workshop Recalls Heartwarming Hugs Bears Due to Risk of Serious Inj Recall 20262026
Frequently asked questions
Is the Bayer HealthCare LLC, of Whippany, New Jersey recall still active?
Yes, this recall is currently active as of April 30, 2026. Do not use or consume the affected product.
How do I know if my Bayer HealthCare LLC, of Whippany, New Jersey is affected?
All units of Bayer HealthCare LLC, of Whippany, New Jersey matching the description above are affected.
What should I do if I have the recalled Bayer HealthCare LLC, of Whippany, New Jersey?
Do not consume or use this product. Return it to the store for a full refund, or dispose of it safely.